Mesolimbic Circuits:
|
Decades of research has demonstrated that mesocorticolimbic dopamine plays a large role in reward seeking behaviors, and these circuits are greatly altered by drugs of abuse. Our laboratory focuses on understanding how these circuits contribute to the long lasting behavioral changes associated with addiction. We have developed a number of approaches to control dopamine release with great precision, using both optogenetics and chemogenetics, allowing us to explore how both tonic and phasic dopamine release patterns alter reward seeking behaviors. We can precisely target the dopamine release in discrete brain regions, such that we can examine how different subregions of the ventral tegmental area (VTA), or specific terminal regions in the striatum, and cortex, contribute to reward related behaviors. We can up- or down-regulate dopamine receptors and other major neurotransmitter components. There are also many new subcircuits being explored that influence different aspects of reward seeking. We use a variety of novel viral approaches to examine how these previously unexplored circuits influence drug seeking behaviors.
|
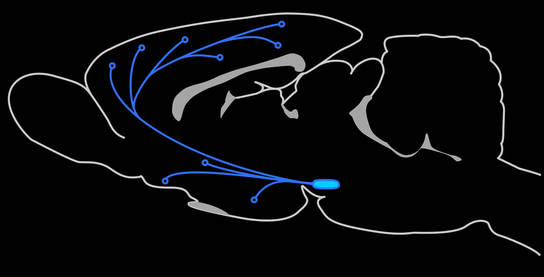
VTA GABA neurons
The VTA is packed with dopamine neurons compromising ~70% of the total population. In addition, ~25% of VTA neurons are thought to be GABAergic interneurons, which provide local inhibitory control over the dopamine neurons. The VTA also contain a smattering of glutamatergic neurons and dopaminergic neurons that co-release either glutamate or GABA. Roughly 5% of the remaining neurons within the VTA are GABA projection neurons, which project to a variety of brain regions, notably the cortex and ventral striatum. These neurons however are not well characterized and their role in mesolimbic function and reward seeking are not well known.
|
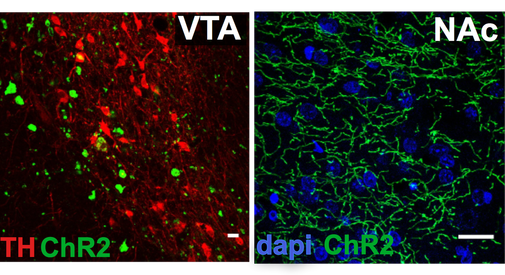
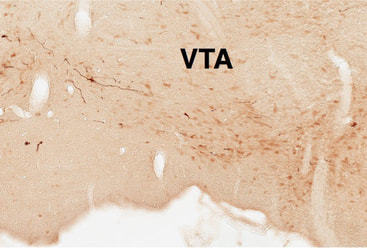
In order to understand the function of VTA neurons and pathways in reward seeking, we must first have a way to independently measure their contribution to behavior. Recently we have developed a novel viral strategy to genetically target VTA GABA neurons and have begun to map their location in the rat VTA and their respective terminal fields. We have found that these neurons project extensively throughout the mesocorticolimbic regions, often in patterns quite distinct from VTA dopamine neurons. One major projection is to the ventral striatum, including the nucleus accumbens and olfactory tubercle. Another major projection is to the prefrontal cortex.
|
Viral targeting of neural subcircuits
We use a variety of viral strategies to selectively deliver genes to specific neuronal subtypes in the rat. We prefer to use rats because we can then study these circuits in sophisticated reward seeking models that have been well validated in rats. Additionally, the larger brains of rats allows us to conduct certain in vivo electrochemical techniques such as fast scan cyclic voltammetry.
Types of viral vectors
|
There are a variety of viral vectors that one can use to manipulate neural circuits in vivo, and the flavor to pick depends upon your needs. The primary viruses used in the brain are herpes simplex virus (HSV), lentivirus, and adeno-associated virus (AAV). Each has unique properties that can be exploited depending on the needs of the experiment. HSV has a quick on/quick off expression pattern, where maximal expression is seen within three days but generally is greatly attenuated to absent at six days post-infusion. This quick peak makes it ideal in situations where a genetic manipulation is being applied to a quickly developing behavior, or when a within subjects design to collect baseline -- experimental -- return to baseline comparisons. However, for behaviors that may take time to develop or require multiple experiments over several days, an AAV is often preferred.
|
Packaging limits - a balancing act with AAVs
Each type of virus has a different external structure (capsid/envelope) of various size, which means there is a general limit to how much genetic material can fit into a given type of virus. HSV can contain approximately 150 kilo bases (kb) of genetic material, lentivirus ~9-10 kb, while AAV is generally restricted to less than 6 kb of genetic information.
One common way to restrict expression of a transgene to a specific neuron is to use a promoter that is active only in that type of neuron. Most generalized viral and "mammalian" promoters (e.g. CMV, CAG, EF1a) are often small but very strong. Neuronal specific promoters can be larger (e.g. NSE), though hSyn is fairly compact. Neuronal subtype specific promoters are often larger still (e.g. 2.5kb TH promoter). At a certain point, it is difficult to fit these larger subtype specific promoters into smaller viruses like AAV. Attempts to shrink subtype specific promoters to minimal promoters often results in loss of specificity. Even if a minimal subtype specific promoter can be identified, it may not be particularly strong. After all, even though a promoter drives expression in a specific type of neuron, it doesn’t mean that neuron naturally makes a lot of that protein. Often subtype specific promoters are relatively weak, and can't drive enough expression to change physiology or behavior..
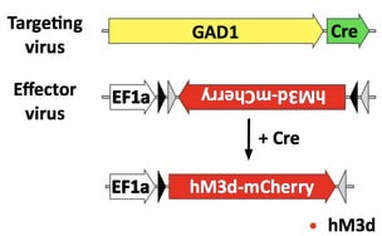
Many labs achieve specificity by using Cre driver lines, these are transgenic mice, and occasionally rats, that produce Cre recombinase driven a specific promoter. These transgenic animals are generated in such a way that promoter size isn’t a factor. Promoter weakness also doesn’t factor in as much, as Cre recombinase is a very efficient enzyme, a little goes a long way. The Cre driver lines can be crossed with a floxed transgenic, or a Cre dependent virus can be infused into the brain.
Targeting neuronal subtypesWe use a combinatorial viral system to drive expression in neuronal subtypes, the first targeting virus uses a large subtype specific promoter to drive expression of Cre recombinase. The second effector virus has a strong generalized promoter, and the gene of interest is housed in a flip excision cassette (I.e. FLEX, DIO) in which the gene of interest is antiparallel to the promoter. However, in the presence of Cre, the cassette is flipped to the proper orientation with the promoter to allow for it’s expression. Even though both viruses transduce neurons at the injection site indiscriminately, Cre will only be made in the targeted neuron, and the FLEX cassette will only be flipped in those neurons. All other neurons will make nonsense transcripts.
|
|
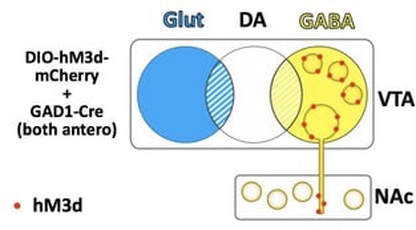
Most AAV serotypes (e.g. AAV2, 5, 8, 10) typically infect cell bodies and are not taken up by terminals, thus by co-infusing the viruses in one brain regions, the gene of interest is overexpressed only in those neurons at the injection sites, and their terminals. Thus to co-infusion into the VTA will target the transgene to VTA GABA interneurons as well as projection neurons.
|
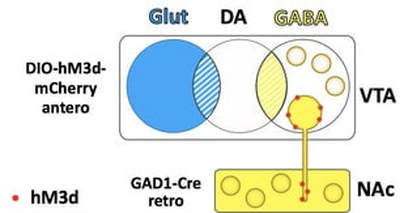
Targeting subtype specific projectionsTo target neuronal subtypes that project to a specific region, we take advantage of viruses that have retrograde properties. In our hands, AAV6 and AAVretro are efficiently taken up by terminals. To target a projection, we infuse the subtype specific Cre retrograde virus in the terminal region, and the FLEX virus in the cell body region. For example, to target GABA neurons that project from the VTA to the NAc, we infuse a FLEX-AAV10 virus into the VTA and a GAD1-Cre-AAV6 virus into the NAc.
|
VTA GABA projections in reward processing
|
The role of VTA GABA projections to the NAc in reward seeking is not clear. Others have shown that VTA GABA activation will decrease sucrose consumption but didn't affect cue-induced anticipatory licking.
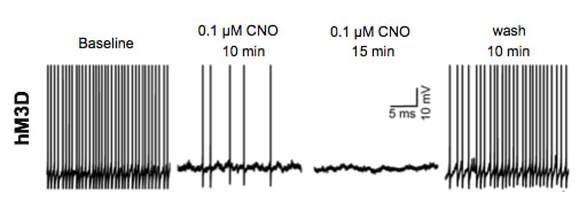
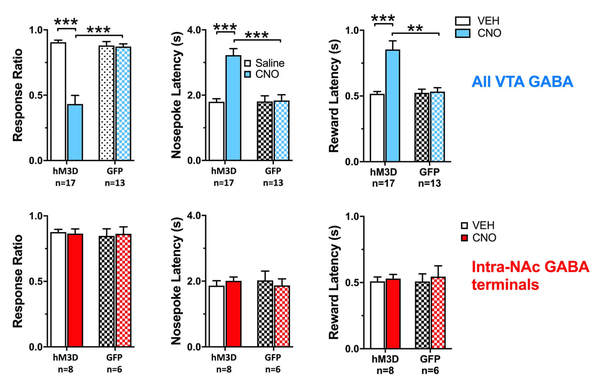
We have recently demonstrated that chemogenetic activation of VTA GABA neurons inhibits responding to reward predictive incentive cues, in an operant model of reward seeking behavior. In this task, rats nosepoke during a distinct, 8-s audiovisual cue, to get a small sucrose reward. We then determine the proportion of cues the rats respond to (response ratio), and the speed with which they respond (nosepoke latency), or collect their reward (reward latency). The latencies are are inversely proportional to their motivation towards the cue or the reward. Activation of the VTA GABA neurons decreases response ratio while increasing the latencies. However, activating the terminals of VTA GABA neurons projecting to the NAc had no impact. This begs the question of what exactly do these mesoaccumbal GABA projections do? We are currently exploring the role of these projections in reward related learning and drug-seeking behaviors. |